Selective CO2-to-CO photoreduction over an orthophosphate semiconductor via the direct Z-scheme heterojunction of Ag3PO4 quantum dots decorated on SnS2 nanosheets†
Abstract
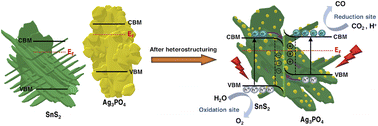
Direct Z-scheme heterojunctions are widely used for photocatalytic water splitting and CO2 reduction due to facilitating well-separated photogenerated charge carriers and spatial isolation of redox reactions. Here, using a facile two-step hydrothermal and ion-exchange method, we uniformly decorate silver orthophosphate (i.e., Ag3PO4) quantum dots with an average characteristic size of ∼10 nm over tin(IV) sulphide (i.e., SnS2) nanosheets to form a 0D/2D heterojunction. The direct Z-scheme mechanism, i.e. charge transport for efficient electron (from SnS2) and hole (from Ag3PO4) recombination, is confirmed by the following experiments: (i) ultraviolet and X-ray photoelectron spectroscopies; (ii) photodeposition of Pt and PbO2 nanoparticles on reduction and oxidation sites, respectively; (iii) in situ X-ray photoelectron spectroscopy; and (iv) electron paramagnetic resonance spectroscopy. Owing to the photoreduction properties of Ag3PO4 with orthophosphate vacancies, Z-scheme charge carrier transfer, and efficient exciton dissociation, an optimized heterojunction shows a high CO2-to-CO reduction yield of 18.3 μmol g−1 h−1 with an illustrious selectivity of ∼95% under light illumination, which is about 3.0 and 47.8 times larger than that of Ag3PO4 and SnS2, respectively. The carbon source for the CO product is verified using a 13CO2 isotopic experiment. Moreover, by tracing the peak at ∼1190 cm−1 in the dark and under light irradiation, in situ diffuse reflectance infrared Fourier transform spectroscopy demonstrates that the CO2 reduction pathway goes through the COOH* intermediate.
- This article is part of the themed collection: Sustainable Energy & Fuels Recent HOT Articles


 Please wait while we load your content...
Please wait while we load your content...