Key difference between transition state stabilization and ground state destabilization: increasing atomic charge densities before or during enzyme–substrate binding†
Abstract
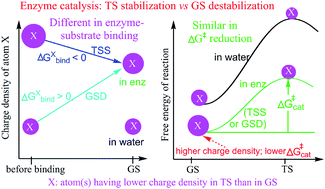
The origin of the enormous catalytic power of enzymes has been extensively studied through experimental and computational approaches. Although precise mechanisms are still subject to much debate, enzymes are thought to catalyze reactions by stabilizing transition states (TSs) or destabilizing ground states (GSs). By exploring the catalysis of various types of enzyme–substrate noncovalent interactions, we found that catalysis by TS stabilization and the catalysis by GS destabilization share common features by reducing the free energy barriers (ΔG‡s) of reactions, but are different in attaining the requirement for ΔG‡ reduction. Irrespective of whether enzymes catalyze reactions by TS stabilization or GS destabilization, they reduce ΔG‡s by enhancing the charge densities of catalytic atoms that experience a reduction in charge density between GSs and TSs. Notably, in TS stabilization, the charge density of catalytic atoms is enhanced prior to enzyme–substrate binding; whereas in GS destabilization, the charge density of catalytic atoms is enhanced during the enzyme–substrate binding. Results show that TS stabilization and GS destabilization are not contradictory to each other and are consistent in reducing the ΔG‡s of reactions. The full mechanism of enzyme catalysis includes the mechanism of reducing ΔG‡ and the mechanism of enhancing atomic charge densities. Our findings may help resolve the debate between TS stabilization and GS destabilization and assist our understanding of catalysis and the design of artificial enzymes.


 Please wait while we load your content...
Please wait while we load your content...