Impact of anion polarizability on ion pairing in microhydrated salt clusters†
Abstract
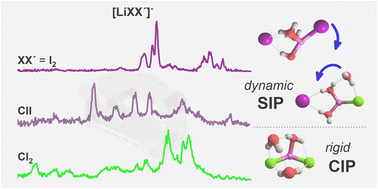
Despite longstanding interest in the mechanism of salt dissolution in aqueous media, a molecular level understanding remains incomplete. Here, cryogenic ion trap vibrational action spectroscopy is combined with electronic structure calculations to track salt hydration in a gas phase model system one water molecule at a time. The infrared photodissociation spectra of microhydrated lithium dihalide anions [LiXX′(H2O)n]− (XX′ = I2, ClI and Cl2; n = 1–3) in the OH stretching region (3800–2800 cm−1) provide a detailed picture of how anion polarizability influences the competition among ion–ion, ion–water and water–water interactions. While exclusively contact ion pairs are observed for n = 1, the formation of solvent-shared ion pairs, identified by markedly red-shifted OH stretching bands (<3200 cm−1), originating from the bridging water molecules, is favored already for n = 2. For n = 3, Li+ reaches its maximum coordination number of four only in [LiI2(H2O)3]−, in accordance with the hard and soft Lewis acid and base principle. Water–water hydrogen bond formation leads to a different solvent-shared ion pair motif in [LiI2(H2O)3]− and network formation even restabilizes the contact ion pair motif in [LiCl2(H2O)3]−. Structural assignments are exclusively possible after the consideration of anharmonic effects. Molecular dynamics simulations confirm that the significance of large amplitude motion (of the water molecules) increases with increasing anion polarizability and that needs to be considered already at cryogenic temperatures.


 Please wait while we load your content...
Please wait while we load your content...