Epitope alteration by small molecules and applications in drug discovery†
Abstract
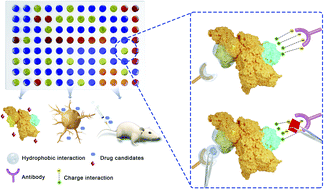
Small molecules and antibodies are normally considered separately in drug discovery, except in the case of covalent conjugates. We unexpectedly discovered several small molecules that could inhibit or enhance antibody–epitope interactions which opens new possibilities in drug discovery and therapeutic modulation of auto-antibodies. We first discovered a small molecule, CRANAD-17, that enhanced the binding of an antibody to amyloid beta (Aβ), one of the major hallmarks of Alzheimer's disease, by stable triplex formation. Next, we found several small molecules that altered antibody–epitope interactions of tau and PD-L1 proteins, demonstrating the generality of this phenomenon. We report a new screening technology for ligand discovery, screening platform based on epitope alteration for drug discovery (SPEED), which is label-free for both the antibody and small molecule. SPEED, applied to an Aβ antibody, led to the discovery of a small molecule, GNF5837, that inhibits Aβ aggregation and another, obatoclax, that binds Aβ plaques and can serve as a fluorescent reporter in brain slices of AD mice. We also found a small molecule that altered the binding between Aβ and auto-antibodies from AD patient serum. SPEED reveals the sensitivity of antibody–epitope interactions to perturbation by small molecules and will have multiple applications in biotechnology and drug discovery.


 Please wait while we load your content...
Please wait while we load your content...