Enantioselective iodolactonization to prepare ε-lactone rings using hypervalent iodine†
Abstract
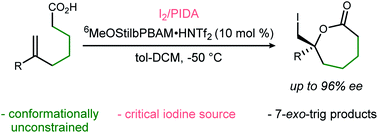
Despite the rapid growth of enantioselective halolactonization reactions in recent years, most are effective only when forming smaller (6,5,4-membered) rings. Seven-membered ε-lactones, are rarely formed with high selectivity, and never without conformational bias. We describe the first highly enantioselective 7-exo-trig iodolactonizations of conformationally unbiased ε-unsaturated carboxylic acids, effected by an unusual combination of a bifunctional BAM catalyst, I2, and I(III) reagent (PhI(OAc)2:PIDA).


 Please wait while we load your content...
Please wait while we load your content...