19F-centred NMR analysis of mono-fluorinated compounds†
Abstract
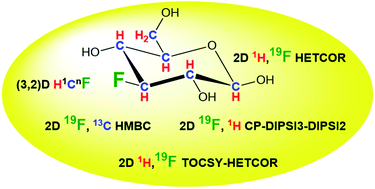
Addressing limitations of the existing NMR techniques for the structure determination of mono-fluorinated compounds, we have developed methodology that uses 19F as the focal point of this process. The proposed 19F-centred NMR analysis consists of a complementary set of broadband, phase-sensitive NMR experiments that utilise the substantial sensitivity of 19F and its far reaching couplings with 1H and 13C to obtain a large number of NMR parameters. The assembled 1H, 13C and 19F chemical shifts, values of JHF, JHH, and JFC coupling constants and the size of 13C induced 19F isotopic shifts constitute a rich source of information that enables structure elucidation of fluorinated moieties and even complete structures of molecules. Here we introduce the methodology, provide a detailed description of each NMR experiment and illustrate their interpretation using 3-fluoro-3-deoxy-D-glucose. This novel approach performs particularly well in the structure elucidation of fluorinated compounds embedded in complex mixtures, eliminating the need for compound separation or use of standards to confirm the structures. It represents a major contribution towards the analysis of fluorinated agrochemicals and (radio)pharmaceuticals at any point during their lifetime, including preparation, use, biotransformation and biodegradation in the environment. The developed methodology can also assist with the investigations of the stability of fluoroorganics and their pharmacokinetics. Studies of reaction mechanisms using fluorinated molecules as convenient reporters of these processes, will also benefit.
- This article is part of the themed collections: Celebrating our 2024 Prizewinners and 2022 RSC Advances Popular Advances Collection


 Please wait while we load your content...
Please wait while we load your content...