A stepwise one-pot synthesis of aliphatic thiols and their derivatives from acrylamides and sulfur†
Abstract
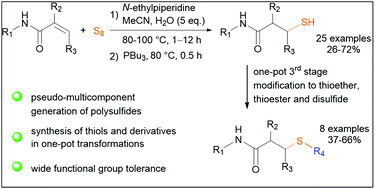
Elemental sulfur enables the convenient formation of C–S bonds and the direct incoporation of S–S bonds. The reactivity of easily accessible electron deficient alkenes towards sulfur, however, is barely disclosed. Herein, we investigated the reactivity of acrylamides with sulfur and eventually developed a new pseudo-multicomponent reaction for the preparation of polysulfides. Sequential one-pot reduction led to diversely substituted thiols. Additional third stage one-pot modifications provided thioethers, unsymmetric disulfide and thioester.


 Please wait while we load your content...
Please wait while we load your content...