Methyl group configuration on acyclic threoninol nucleic acids (aTNAs) impacts supramolecular properties†
Abstract
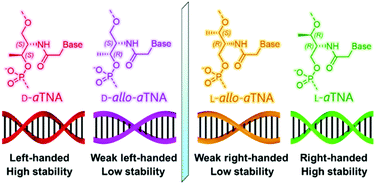
We have synthesized acyclic allo-threoninol nucleic acids (allo-aTNAs), artificial xeno-nucleic acids (XNAs) that are diastereomers of acyclic threoninol nucleic acids (aTNAs), and have investigated their supramolecular properties. The allo-aTNAs formed homo-duplexes in an antiparallel manner but with lower thermal stability than DNA, whereas aTNAs formed extremely stable homo-duplexes. The allo-aTNAs formed duplexes with complementary aTNAs and serinol nucleic acid (SNA). The affinities of L-allo-aTNA were the highest for L-aTNA and the lowest for D-aTNA, with SNA being intermediate. The affinities of D-allo-aTNA were the reverse. Circular dichroism measurements revealed that L- and D-allo-aTNAs had weak right-handed and left-handed helicities, respectively. The weak helicity of allo-aTNAs likely explains the poor chiral discrimination of these XNAs, which is in contrast to aTNAs that have strong helical orthogonality. Energy-minimized structures of L-allo-aTNA/RNA and L-allo-aTNA/L-allo-aTNA indicated that the methyl group on the allo-aTNA strand is unfavourable for duplex formation. In contrast, the methyl group on L-aTNA likely stabilizes the duplex structure via hydrophobic effects and van der Waals interactions. Thus, the configuration of the methyl group on the XNA scaffold had an unexpectedly large impact on the hybridization ability and structure.


 Please wait while we load your content...
Please wait while we load your content...