Non-invasive transferrin targeted nanovesicles sensitize resistant glioblastoma multiforme tumors and improve survival in orthotopic mouse models†
Abstract
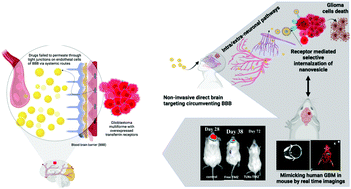
The blood–brain barrier (BBB) and tumor heterogeneity have resulted in abysmally poor clinical outcomes in glioblastoma (GBM) with the standard therapeutic regimen. Despite several anti-glioma drug delivery strategies, the lack of adequate chemotherapeutic bioavailability in gliomas has led to a suboptimal therapeutic gain in terms of improvement in survival and increased systemic toxicities. This has paved the way for designing highly specific and non-invasive drug delivery approaches for treating GBM. The intranasal (IN) route is one such delivery strategy that has the potential to reach the brain parenchyma by circumventing the BBB. We recently showed that in situ hydrogel embedded with miltefosine (HePc, proapoptotic anti-tumor agent) and temozolomide (TMZ, DNA methylating agent) loaded targeted nanovesicles prevented tumor relapses in orthotopic GBM mouse models. In this study, we specifically investigated the potential of a non-invasive IN route of TMZ delivered from lipid nanovesicles (LNs) decorated with surface transferrin (Tf) and co-encapsulated with HePc to reach the brain by circumventing the BBB in glioma bearing mice. The targeted nanovesicles (228.3 ± 10 nm, −41.7 ± 4 mV) exhibited mucoadhesiveness with 2% w/v mucin suggesting their potential to increase brain drug bioavailability after IN administration. The optimized TLNs had controlled, tunable and significantly different release kinetics in simulated cerebrospinal fluid and simulated nasal fluid demonstrating efficient release of the payload upon reaching the brain. Drug synergy (combination index, 0.7) showed a 6.4-fold enhanced cytotoxicity against resistant U87MG cells compared to free drugs. In vivo gamma scintigraphy of 99mTc labeled LNs showed 500- and 280-fold increased brain concentration post 18 h of treatment. The efficacy of the TLNs increased by 1.8-fold in terms of survival of tumor-bearing mice compared to free drugs. These findings suggested that targeted drug synergy has the potential to intranasally deliver a high therapeutic dose of the chemotherapy agent (TMZ) and could serve as a platform for future clinical application.


 Please wait while we load your content...
Please wait while we load your content...