Synergism and properties of binary mixtures based on an arginine dodecyl ester surfactant
Abstract
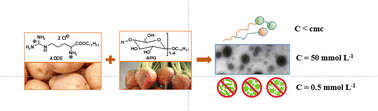
The properties of the binary mixtures of a new cationic amino acid surfactant arginine dihydrochloride dodecyl ester (ADDE) and alkyl polyglycosides (APGs) were studied systematically by evaluating surface tension, conductivity, dynamic light scattering (DLS), transmission electron microscopy (TEM), biological and cytotoxic activity measurements. Higher surface activity and more diverse self-assembly behaviors for the mixed system (ADDE/APG) compared to single systems (ADDE or APG alone) were seen due to the synergistic effects between ADDE and APG. The interaction and micelle thermodynamic parameters of ADDE/APG mixtures show strong intermolecular interaction, both in the mixed micelles and at the gas/liquid interface, and indicate that the micellization is an enthalpy-driven process. At the total solution concentration of 50 mmol L−1 and higher mole fraction of ADDE (αADDE ≥ 0.5), primarily large aggregates are formed according to DLS results with typically spherical vesicles of about 200 nm in diameter as shown by TEM. The experimental results also show that the antimicrobial activities of ADDE/APG mixtures are outstanding at a low concentration of 0.5 mmol L−1 and are attributed to ADDE containing amino and guanidinium cationic groups within the same molecule. And the hemolytic activity results of ADDE/APG mixtures clearly indicate that the maximum blood compatible concentration was under 0.5 mmol L−1. This research will be beneficial for broadening the application field of amino acid surfactant ADDE.


 Please wait while we load your content...
Please wait while we load your content...