Ferrocene functionalized enantiomerically pure Schiff bases and their Zn(ii) and Pd(ii) complexes: a spectroscopic, crystallographic, electrochemical and computational investigation†
Abstract
In this contribution, we describe the synthesis, spectroscopic and structural characterization, as well as the electrochemical behavior of a series of four chiral ferrocene-functionalized Schiff base compounds derived from enantiomerically pure (1R,2R)-(−)-1,2-diaminocyclohexane. The ferrocene enaminones 3 and 4 were obtained upon monocondensation of ferrocenoylacetone Fc–C(![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O)CH2C(
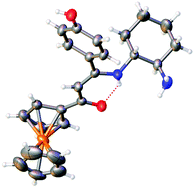
O)CH2C(![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O)R (R = CH3, 1; 4-C6H4OH, 2; Fc = (η5-C5H5)Fe(η5-C5H4)) with one amino group of the primary diamine. The two bimetallic complexes 5 and 6 were prepared via a one-pot three-components template procedure involving half-unit 3, 4-methoxysalicylaldehyde and tetrahydrated zinc(II) nitrate for 5, and half-unit 4, 4-formyl-3-hydroxybenzoic acid and palladium(II) acetate for 6. All the syntheses were carried out in refluxing ethanol and compounds 3–6 were isolated as thermally stable, air and moisture compatible colored solids in 71–83% yields. They were characterized by several analytical (EA, ESI-MS) and spectroscopic methods (1H/13C NMR, FT-IR, UV-vis), including the molecular structures of 3 and 4 that were unequivocally established by single crystal X-ray diffraction analyses. Both crystallize in the orthorhombic non-centrosymmetric space group P212121 with two (R,R)-(−)-chiral carbon atoms in the structures. Cyclic voltammetry showed that the greatest anodic shifts (270 mV) of the ferrocenyl Fe(II)/Fe(III) redox potentials are observed for the D-π-A push–pull derivative 6. Beyond experimental characterization, DFT- and TDDFT-based theoretical analyses of the new chiral ferrocene-containing Schiff base complexes allow interpretation of their observed spectroscopic, structural and electronic features.
O)R (R = CH3, 1; 4-C6H4OH, 2; Fc = (η5-C5H5)Fe(η5-C5H4)) with one amino group of the primary diamine. The two bimetallic complexes 5 and 6 were prepared via a one-pot three-components template procedure involving half-unit 3, 4-methoxysalicylaldehyde and tetrahydrated zinc(II) nitrate for 5, and half-unit 4, 4-formyl-3-hydroxybenzoic acid and palladium(II) acetate for 6. All the syntheses were carried out in refluxing ethanol and compounds 3–6 were isolated as thermally stable, air and moisture compatible colored solids in 71–83% yields. They were characterized by several analytical (EA, ESI-MS) and spectroscopic methods (1H/13C NMR, FT-IR, UV-vis), including the molecular structures of 3 and 4 that were unequivocally established by single crystal X-ray diffraction analyses. Both crystallize in the orthorhombic non-centrosymmetric space group P212121 with two (R,R)-(−)-chiral carbon atoms in the structures. Cyclic voltammetry showed that the greatest anodic shifts (270 mV) of the ferrocenyl Fe(II)/Fe(III) redox potentials are observed for the D-π-A push–pull derivative 6. Beyond experimental characterization, DFT- and TDDFT-based theoretical analyses of the new chiral ferrocene-containing Schiff base complexes allow interpretation of their observed spectroscopic, structural and electronic features.


 Please wait while we load your content...
Please wait while we load your content...