The effect of side chains on the complex formation processes of N-terminally free hexapeptides containing C-terminal cysteinyl functions†
Abstract
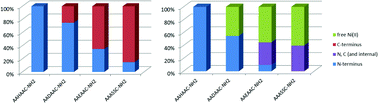
Cysteine containing ligands with free amino- and amidated C-termini were studied and their nickel(II), zinc(II) and cadmium(II) complexes were described in the aspect of their thermodynamic stability and structure, including the preference of the metal ions toward the two anchoring groups. Comparing the investigated ligands, AAASSC-NH2 and AAEAAC-NH2, with the previously studied ones, AAHAAC-NH2 and AADAAC-NH2, it can be stated that the ratio of coordination isomers of nickel(II) complexes can be altered by introducing strongly, weakly or non-coordinating internal amino acid residues. Metal binding priority of the C-terminal binding site enhances in an order of AAASSC-NH2 > AAEAAC-NH2 > AADAAC-NH2 > AAHAAC-NH2, forming an (N−,N−,N−,S−) fused chelate system at high pH. Under neutral conditions, in contrast with the former investigated ligands, a macrochelate structure is present in the NiL species without the involvement of amide nitrogen atoms, and in the case of AAEAAC-NH2 the γ-carboxylate function of glutamic acid residue contributes to the stability of this complex. The formation of zinc(II) complexes occurred with the stoichiometry of ZnHL and ZnL, the latter containing the amino function in the coordination sphere, too, besides the thiolate (and carboxylate) groups. This finding is in good agreement with the ligand containing aspartic acid, but is in contrast with the strongly coordinating histidine peptide, where bis(ligand) zinc(II) species were also formed. Bis(ligand) complex formation was more facilitated in the case of cadmium(II) ions, except for AAASSC-NH2. All the other three ligands were able to form CdH2L2, CdHL2 and CdL2 species, indicating that an internal, even only weakly coordinating side chain enables the coordination of two ligands to a metal ion due to the tridentate binding of the peptide.


 Please wait while we load your content...
Please wait while we load your content...