Substituent effects on the mechanochemical response of zinc dialkyldithiophosphate†
Abstract
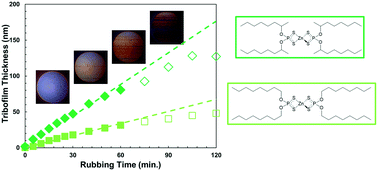
Mechanochemistry is known to play a key role in the function of some lubricant additives, such as the tribofilm growth of zinc dialkyldithiophosphate (ZDDP). This raises the intriguing possibility of tailoring the mechanochemical response of additives by modifying their alkyl substituents. Here, we study the tribofilm formation rate of ZDDPs containing several different alkyl groups on steel surfaces from a high-friction base oil. We use macroscale tribometer experiments under full-film elastohydrodynamic lubrication conditions to enable careful control of the temperature and stress during tribofilm growth. We show how the chain length and the presence of branches or bulky cycloaliphatic groups can lead to large differences in the temperature- and stress-dependencies of the tribofilm formation rate, which can be explained through variations in packing density, steric hindrance, and stress transmission efficiency. Our rate data are successfully fitted using the Bell model; a simple modification of the Arrhenius equation that is commonly employed to model the kinetics of mechanochemical processes. Using this model, we observe large differences in the activation energy, pre-exponential factor, and activation volume for the various ZDDPs. Our findings show how structure–performance relationships can be identified for lubricant additives, which may be useful to optimise their molecular structure.


 Please wait while we load your content...
Please wait while we load your content...