Combined chemoenzymatic strategy for sustainable continuous synthesis of the natural product hordenine†
Abstract
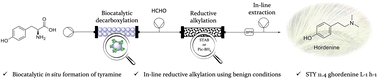
To improve sustainability, safety and cost-efficiency of synthetic methodologies, biocatalysis can be a helpful ally. In this work, a novel chemoenzymatic stategy ensures the rapid synthesis of hordenine, a valuable phenolic phytochemical under mild working conditions. In a two-step cascade, the immobilized tyrosine decarboxylase from Lactobacillus brevis (LbTDC) is here coupled with the chemical reductive amination of tyramine. Starting from the abundant and cost-effective amino acid L-tyrosine, the complete conversion to hordenine is achieved in less than 5 minutes residence time in a fully-automated continuous flow system. Compared to the metal-catalyzed N,N-dimethylation of tyramine, this biocatalytic approach reduces the process environmental impact and improves its STY to 2.68 g L−1 h−1.


 Please wait while we load your content...
Please wait while we load your content...