A sobering examination of the feasibility of aqueous aluminum batteries†
Abstract
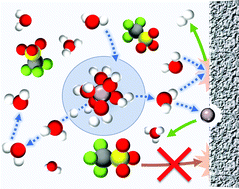
Aqueous aluminum (Al) batteries are posited to be a cheap and energy dense alternative to conventional Li-ion chemistries, but an aqueous electrolyte mediating trivalent aluminum cations (Al3+) warrants greater scrutiny. This study provides a rigorous examination of aqueous Al electrolytes, with the first compelling evidence for a dynamic octahedral solvation structure around the Al3+, without Al–OTf contact ion pairs, even at high concentrations. This solvation behavior and the concomitant, transient electrostatic hydrolysis of Al–OH2 ligands contrasts strongly with previously reported water-in-salt electrolytes, and occurs due to the high charge density of the Lewis acidic Al3+. Nuclear magnetic resonance spectroscopy and other physicochemical measurements quantitatively reveal how species activity evolves with concentration and temperature. This new understanding exposes practical concerns related to the corrosiveness of the acidic aqueous solutions, the degree of hydration of aluminum trifluoromethanesulfonate (Al(OTf)3) salt, and the grossly insufficient reductive stability of the proposed electrolytes (>1 V between HER onset and Al3+/Al). Collectively, these factors constitute multiple fundamental barriers to the feasibility of rechargeable aqueous Al batteries.


 Please wait while we load your content...
Please wait while we load your content...