Migration-assisted, moisture gradient process for ultrafast, continuous CO2 capture from dilute sources at ambient conditions†
Abstract
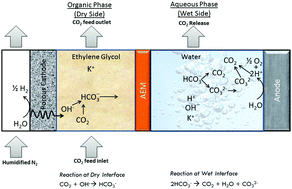
The current industrially relevant CO2 capture technologies require high operating and capital costs or are energy-intensive, rendering them inefficient and unsustainable for long-term use. The higher operating cost is primarily due to the energy-intense swing of pH- or temperature-dependent equilibrium of CO32− and HCO3− to capture CO2. CO2 can be captured in an aqueous solution with OH− to convert it into HCO3−, which can be turned back into CO32− and CO2 using either temperature or pH swing. This captured CO2 can be either sequestered or chemically transformed into a usable product. The challenges with this technology are the limited carbon capacity of the aqueous solvent system, higher energy requirement for solvent regeneration, and loss of solvent through evaporation. The water-dependent equilibrium of CO2 with CO32− and HCO3− can be a cost-effective alternative to the existing technologies. Here, we exploit this phenomenon by establishing a water gradient across an anion exchange membrane that separates organic solvent and an aqueous solution. The organic solution contains ethylene glycol saturated with KOH that captures CO2 from dilute sources such as air or flue gas and produces a high concentration of HCO3− that migrates towards the aqueous side, where HCO3− converts back to CO2 for its end-use. The current efficiency of CO2 capture is nearly 100%, with a tunable flux CO2 capture flux that is controlled by the applied current density. The water-dependent CO2 capture and release kinetics are measured using in situ Fourier-transform infrared spectroscopy and validated using density functional theory. A detailed transport model confirms the CO2 capture, transport, and release mechanism for varying water concentration in the organic solution, membrane thickness, and the applied current. Machine learning model predicts the performance curve for this process that relates CO2 saturation in aqueous solution to the CO2 capture efficiency, relative humidity of flue gas, and current density. The validated prototype of the electrodialysis system showed an unprecedented CO2 capture flux of 2.3 mmol m−2 s−1, which is 100 times higher than the state-of-the-art existing CO2 capture technologies. The technoeconomic analysis predicts ∼$145 per ton of CO2 for 1000 ton per hour of CO2 capture capacity.


 Please wait while we load your content...
Please wait while we load your content...
