Isoelectronic Pt(ii) complexes of cyclometalating C^N^N ligands with phenyl/(benzo)thiophenyl and pyridyl/(benzo)thiazolyl moieties†
Abstract
A series of cyclometalated Pt(II) complexes [Pt(C^N^N)X] (X = Cl, C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) CPh, C
CPh, C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) CC6F5) was synthesised from the protoligands HC^N^N containing either phenyl (ph), naphthyl (na) or (benzo)thiophenyl (b(th)) C aryl functions and either pyridyl (py) or (benzo)thiazolyl ((b)tz) peripheral N units, alongside the central 4-phenyl-pyridyl (ppy) or tBu2-phenyl-pyridyl (tbppy) N group. Depending on the combination of the peripheral N or C aryl building blocks, these square planar complexes reveal very different electrochemical, UV-vis absorption and emission behaviour. The reversible reductions shift anodically along the series th/py < ph/tz ≈ th/tz < ph/btz while the irreversible oxidations shift cathodically along the series Cl ≈ C
CC6F5) was synthesised from the protoligands HC^N^N containing either phenyl (ph), naphthyl (na) or (benzo)thiophenyl (b(th)) C aryl functions and either pyridyl (py) or (benzo)thiazolyl ((b)tz) peripheral N units, alongside the central 4-phenyl-pyridyl (ppy) or tBu2-phenyl-pyridyl (tbppy) N group. Depending on the combination of the peripheral N or C aryl building blocks, these square planar complexes reveal very different electrochemical, UV-vis absorption and emission behaviour. The reversible reductions shift anodically along the series th/py < ph/tz ≈ th/tz < ph/btz while the irreversible oxidations shift cathodically along the series Cl ≈ C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) CC6F5 < C
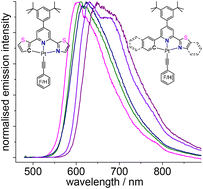
CC6F5 < C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) CPh. Similar trends were observed for the long-wavelength UV-vis absorption and photoluminescence properties. The emission maxima range from 605 to 675 nm at 298 K in CH2Cl2 solution and from 555 to 655 nm at 77 K in glassy frozen CH2Cl2/MeOH matrices. Large differences in amplitude-weighted average lifetimes τav (up to 0.9 μs at 298 K, up to 12 μs at 77 K) and photoluminescence quantum yields ΦL (up to 0.15 at 298 K and up to 0.82 at 77 K) were found. TD-DFT calculations showed that the decomposition of the triplet excited states into LC (π–π*, centred in the individual parts of the C^N^N ligand) and LLCT (π–π*, between the individual parts of the C^N^N ligand + X–π* from coligand to C^N^N) contributions for the ligand-centred states as well as MLCT (dPt-to-π*C^N^N) and LMCT (pCl or πCCR-to-dPt) character for the charge-transfer states involving the metal is beneficial to assess the participation of the individual heteroaryl groups of the C^N^N ligands. In view of the modular synthesis of these ligands, this will allow the realisation of tailor-made Pt(II) triplet emitters in future work.
CPh. Similar trends were observed for the long-wavelength UV-vis absorption and photoluminescence properties. The emission maxima range from 605 to 675 nm at 298 K in CH2Cl2 solution and from 555 to 655 nm at 77 K in glassy frozen CH2Cl2/MeOH matrices. Large differences in amplitude-weighted average lifetimes τav (up to 0.9 μs at 298 K, up to 12 μs at 77 K) and photoluminescence quantum yields ΦL (up to 0.15 at 298 K and up to 0.82 at 77 K) were found. TD-DFT calculations showed that the decomposition of the triplet excited states into LC (π–π*, centred in the individual parts of the C^N^N ligand) and LLCT (π–π*, between the individual parts of the C^N^N ligand + X–π* from coligand to C^N^N) contributions for the ligand-centred states as well as MLCT (dPt-to-π*C^N^N) and LMCT (pCl or πCCR-to-dPt) character for the charge-transfer states involving the metal is beneficial to assess the participation of the individual heteroaryl groups of the C^N^N ligands. In view of the modular synthesis of these ligands, this will allow the realisation of tailor-made Pt(II) triplet emitters in future work.


 Please wait while we load your content...
Please wait while we load your content...