Non-innocent cyanido ligands: tetracyanidoferrate(–ii) as carbonyl copycat†
Abstract
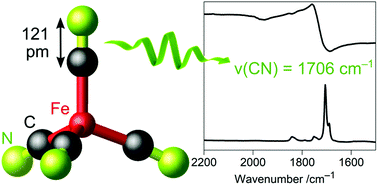
While a negative oxidation state occurs rarely for metals in general, this is commonly known for metal carbonyl anions, i.e. carbonyl metalates. Although CO and CN− are isoelectronic, cyanidometalates usually do not exhibit metal centers with negative oxidation states. However, we report on the electron-rich tetrahedral tetracyanidoferrate(–II) anion [Fe(CN)4]6−, which was stabilized in (Sr3N)2[Fe(CN)4] (space group R3c, a = 702.12(2) pm, c = 4155.5(2) pm). Microcrystalline powders were synthesized by a solid-state route, single crystals were obtained from Na metal flux. In comparison to classical cyanidometalates, C–N distances are longer and stretching frequencies are lower as indicated by X-ray diffraction, IR and Raman spectroscopy. Weak C–N, strong Fe–C bonds as well as the anion geometry resemble the isoelectronic tetrahedral carbonyl ferrate [Fe(CO)4]2−. 57Fe Mössbauer spectroscopic measurements reveal a negative isomer shift in agreement with substantially delocalized d electrons due to strong π back-bonding. These results point to a very similar bonding situation of both 18e tetracyanido and tetracarbonyl ferrates including non-innocent redox-active ligands and a d10 closed shell configuration on iron. Hereby, new tetracyanidoferrate(–II) provides a missing link for a more in-depth understanding of the chemical bonding trends of highly-reduced cyanidometalates in the quest for even higher reduced transition metals in this exceptional class of compounds.
- This article is part of the themed collection: Open Access in Dalton Transactions


 Please wait while we load your content...
Please wait while we load your content...