Merging liquid crystalline self-assembly and linear optical properties of merocyanines via tailored donor units†
Abstract
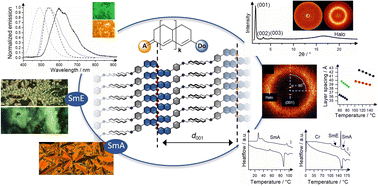
Aiming at merocyanine dyes with good linear optical and self-assembly properties, a series of rigid mono-, bi- and tricyclic merocyanines with O- and N-donor units as well as keto or malodinitrile acceptor units was prepared by a convergent approach. With particular focus on tailoring the donor unit, a selection of appropriate derivatives was investigated with respect to their dye properties in solution and in the bulk (UV/Vis, fluorescence, temperature-dependent fluorescence, lifetime). Determination of fluorescence quantum yields revealed the importance of the donor unit and the chromophore size. Larger chromophores and N-donors were beneficial for strong emission in solution, whereas small chromophores and O-donors favored emission in the solid state. To rationalize the different optical properties depending on their donor unit, density functional theory (DFT) calculations were performed. Liquid crystalline derivatives were additionally studied by optical polarization microscopy, differential scanning calorimetry, and X-ray diffraction experiments. For merocyanines with O-donor, fluorinated side chains were mandatory to get stable enantiotropic SmA phases regardless of chromophore size, side chain lengths or acceptor unit. Increased mesophase widths (up to 134 K) were observed upon increasing the chromophore lengths, chain lengths (up to C12) and F/C ratio in the side chain. On the other hand, merocyanines with N-donor and keto acceptor showed enantiotropic SmA phases in the presence of simple alkoxy side chains. The tricyclic merocyanine with N-donor shows an additional SmE phase at lower temperatures. The results revealed the importance of the donor unit to balance optical and mesomorphic properties in merocynanines.


 Please wait while we load your content...
Please wait while we load your content...