Reaction-driven selective CO2 hydrogenation to formic acid on Pd(111)†
Abstract
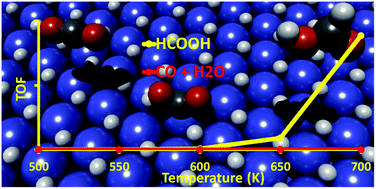
Conversion of CO2 to useful fuels and chemicals has gained great attention in the past decades; yet the challenge persists due to the inert nature of CO2 and the wide range of products formed. Pd-based catalysts are extensively studied to facilitate CO2 hydrogenation to methanol via a reverse water gas shift (rWGS) pathway or formate pathway where formic acid may serve as an intermediate species. Here, we report the selective production of formic acid on the stable Pd(111) surface phase under CO2 hydrogenation conditions, which is fully covered by chemisorbed hydrogen, using combined Density Functional Theory (DFT) and Kinetic Monte Carlo (KMC) simulations. The results show that with the full coverage of hydrogen, instead of producing methanol as reported for Pd(111), the CO2 activation is highly selective to formic acid via a multi-step process involving the carboxyl intermediate. The high formic acid selectivity is associated with surface hydrogen species on Pd(111), which not only acts as a hydrogen reservoir to facilitate the hydrogenation steps, but also enables the formation of confined vacancy sites to facilitate the production and removal of formic acid. Our study highlights the importance of reactive environments, which can transform the surface structures and thus tune the activity/selectivity of catalysts.


 Please wait while we load your content...
Please wait while we load your content...
