An optimal acquisition scheme for Q-band EPR distance measurements using Cu2+-based protein labels†
Abstract
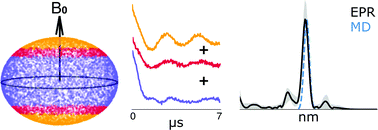
Recent advances in site-directed Cu2+ labeling of proteins and nucleic acids have added an attractive new methodology to measure the structure-function relationship in biomolecules. Despite the promise, accessing the higher sensitivity of Q-band Double Electron Electron Resonance (DEER) has been challenging for Cu2+ labels designed for proteins. Q-band DEER experiments on this label typically require many measurements at different magnetic fields, since the pulses can excite only a few orientations at a given magnetic field. Herein, we analyze such orientational effects through simulations and show that three DEER measurements, at strategically selected magnetic fields, are generally sufficient to acquire an orientational-averaged DEER time trace for this spin label at Q-band. The modeling results are experimentally verified on Cu2+ labeled human glutathione S-transferase (hGSTA1-1). The DEER distance distribution measured at the Q-band shows good agreement with the distance distribution sampled by molecular dynamics (MD) simulations and X-band experiments. The concordance of MD sampled distances and experimentally measured distances adds growing evidence that MD simulations can accurately predict distances for the Cu2+ labels, which remains a key bottleneck for the commonly used nitroxide label. In all, this minimal collection scheme reduces data collection time by as much as six-fold and is generally applicable to many octahedrally coordinated Cu2+ systems. Furthermore, the concepts presented here may be applied to other metals and pulsed EPR experiments.


 Please wait while we load your content...
Please wait while we load your content...
