Excited state dynamics of protonated dopamine: hydration and conformation effects†
Abstract
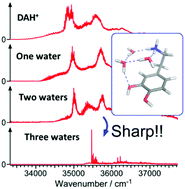
Electronic and vibrational spectroscopy in a cryogenic ion trap has been applied to protonated dopamine water clusters and assigned with the help of quantum chemistry calculations performed in the ground and electronic excited states. A dramatic hydration effect is observed when dopamine is solvated by three water molecules. The broad electronic spectra recorded for the bare and small water clusters containing protonated dopamine turn to sharp, well-resolved vibronic transitions in the 1–3 complex. This reflects the change induced by hydration in the photodynamics of protonated dopamine which is initially controlled by an excited state proton transfer (ESPT) reaction from the ammonium group toward the catechol ring. Interestingly, conformer selectivity is revealed in the 1–3 complex which shows two low lying energy conformers for which the ESPT reaction is prevented or not depending on the H-bond network formed between the dopamine and water molecules.


 Please wait while we load your content...
Please wait while we load your content...