Abstract
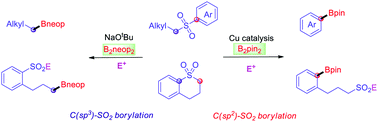
A Cu-catalysed borylation of aryl alkyl sulfones was developed for the high yield synthesis of versatile arylboronic esters using a readily prepared NHC–Cu catalyst. In addition, the selective cleavage of either alkyl(C)–sulfonyl or aryl(C)–sulfonyl bonds of a cyclic sulfone via Cu-free or Cu-mediated processes generates the corresponding sulfinate salts, which can be further derivatised to provide sulfonyl-containing boronate esters, such as sulfones and sulfonyl fluorides.
- This article is part of the themed collection: Boron Chemistry in the 21st Century: From Synthetic Curiosities to Functional Molecules


 Please wait while we load your content...
Please wait while we load your content...