Mapping the time dependent DNA fragmentation caused by doxorubicin loaded on PEGylated carbogenic nanodots using fluorescence lifetime imaging and superresolution microscopy†
Abstract
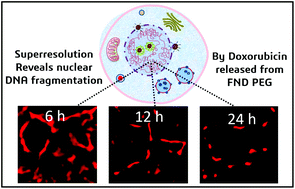
Doxorubicin is an anthracycline drug most commonly used in cancer therapy. It intercalates with the nuclear DNA and induces toxicity by causing DNA breaks and histone eviction. However, the kinetics of its action on the nucleus has not been mapped effectively. This study shows successful PEGylation and DOX loading through π–π interaction onto carbogenic fluorescent nanodots (FNDs), which have an affinity for the nucleolus. Then the drug release from the nanoparticle and its action on the nuclear environment were aptly mapped using both fluorescence lifetime imaging and superresolution radial fluctuation (SRRF) techniques. Here for the first time, the nuclear degradation kinetics caused by the released DOX from the FNDs as a result of DNA double-strand breaks and histone eviction was visualized. This led to the observation of decreasing length, breadth, and complex structure of the nuclear clusters from 6 h to 24 h, resulting in isolated cluster visualization. However, the superresolution images for free DOX and untreated cells reveal no such drastic effects at the same concentration and time points, unlike DOX loaded particles.


 Please wait while we load your content...
Please wait while we load your content...