The effect of temperature and time on the properties of 2D Cs2ZnBr4 perovskite nanocrystals and their application in a Schottky barrier device†
Abstract
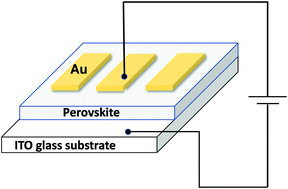
2D hybrid perovskites are promising materials for solar cell applications, in particular, cesium-based perovskite nanocrystals as they offer the stability that is absent in organic–inorganic perovskites. However, the most commonly studied are the lead halides and the toxicity of lead has come under much scrutiny. Cs2ZnBr4 is a less studied Cs-based perovskite that is less toxic than lead halides, offers higher stability than the organic–inorganic perovskites, and has optoelectronic properties suitable for application in solar cells. Herein, we report the colloidal synthesis of Cs2ZnBr4 nanocrystals for the first time. We studied their properties by varying the temperature and time. We then used these resultant nanocrystals in a Schottky diode. Varying the temperature and time resulted in a change in particle size and morphology. These inevitably resulted in different optical properties. The optimum temperature and time were 160 °C and 1 min. The surface chemistry of the nanocrystals was studied using XPS, FT-IR, and NMR techniques. This confirmed that the nanocrystals were capped by oleylamine. The nanocrystals were still stable for 25 days, as confirmed by XRD and TGA. From the Schottky barrier diode, the ideality factor values, barrier heights, and series resistances were determined using the traditional thermionic emission theory of a Schottky barrier, the modified Cheung's and Norde's functions. Notably, high ideality factor values were obtained suggesting poor interface properties. Nevertheless, the optical properties, saturation current and the rectification of the I–V curve suggest that Cs2ZnBr4 nanocrystals are good candidates for use as hole or electron transporting layers in solar cells.


 Please wait while we load your content...
Please wait while we load your content...