Selective and stoichiometric incorporation of ATP by self-assembling amyloid fibrils†
Abstract
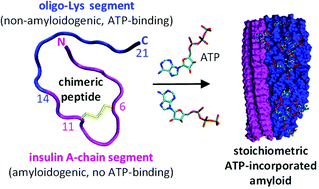
ATP acts as a biological hydrotrope preventing protein aggregation. Here, we report a novel chimeric peptide, ACC1–13K8, with an unusual capacity to bind and incorporate ATP while self-assembling into amyloid fibrils. The amino acid sequence combines a highly amyloidogenic segment of insulin's A-chain (ACC1–13) and octalysine (K8). Fibrillization requires binding 2 ATP molecules per ACC1–13K8 monomer and is not triggered by adenosine di- and monophosphates (ADP, AMP). Infrared and CD spectra and AFM-based morphological analysis reveal tight and orderly entrapment of ATP within superstructural hybrid peptide–ATP fibrils. The incorporation of ATP is an emergent property of ACC1–13K8 not observed for ACC1–13 and K8 segments separately. We demonstrate how new functionalities (e.g. ATP storage) emerge from synergistic coupling of amyloidogenic segments with non-amyloidogenic peptide ligands, and suggest that ATP's role in protein misfolding is more nuanced than previously assumed.

 Please wait while we load your content...
Please wait while we load your content...