Development and demonstration of functionalized inorganic–organic hybrid copper phosphate nanoflowers for mimicking the oxidative reactions of metalloenzymes by working as a nanozyme†
Abstract
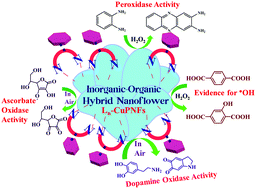
Copper phosphate nanoflowers (CuPNFs) have been synthesized in the presence of different aromatic phenanthroline derivatives (Ln), leading to inorganic–organic hybrid materials (Ln-CuPNFs). Studies revealed that the morphology of nanoflowers varies as a function of the aromatic moiety present in the derivative, Ln (where ‘n’ corresponds to phenyl, naphthyl, anthracenyl, and pyrenyl) used for coating the nanomaterial. Other noticeable changes were the increase in the size of the flower by ∼2–3 fold in the presence of these derivatives. In the absence of such aromatic phenanthroline derivatives, i.e., the use of 1,10-phenanthroline-5-amine did not induce the formation of nanoflowers, suggesting that the organic derivatization used in the present study stabilizes the nanoflower structure. Nanoflowers were characterized using X-ray diffraction, Brunauer–Emmett–Teller (BET) isotherm, X-ray photoelectron spectroscopy, Raman and Infrared spectroscopy, scanning electron microscopy, and transmission electron microscopy, thus covering a range of diffraction, spectroscopy, and microscopy techniques. Nanoflowers, Ln-CuPNFs, have been demonstrated for the oxidative reactions mimicking copper metalloenzymes in the presence and absence of hydrogen peroxide using different substrates. Thus, hybrid Ln-CuPNFs mediate the complete oxidation of o-phenylenediamine, dopamine, ascorbate oxidase, and terephthalic acid without causing much change in the morphology of the hybrid nanoflower material and with the retention of the activity supporting the hybrid as an acceptable enzyme mimicking material. Oxidation is mediated through hydroxyl radical formation and the order of the oxidative activity is pyrenyl > anthracenyl > naphthyl > phenyl for the inorganic–organic hybrid nanoflowers. The copper complex of pyrenyl-appended phenanthroline derivative also showed similar biomimetic activity.


 Please wait while we load your content...
Please wait while we load your content...