Covalently encapsulating sulfur chains into carbon-rich nanomaterials towards high-capacity and high-rate sodium-ion storage†
Abstract
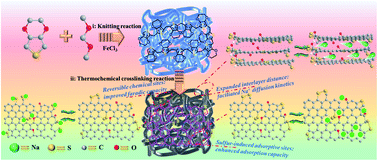
Synchronously achieving high-capacity and high-rate sodium-ion storage is critical for large-scale energy stationary applications of sodium-ion batteries (SIB). Here we present a facile bottom-up strategy to covalently encapsulate sulfur chains into carbon-rich nanomaterials by knitting 3,4-ethylenedioxythiophene (EDOT) with external crosslinking reactants. Using a series of ex situ characterization studies, the synergistic effects of the covalent sulfur species (21.33 wt%) are demonstrated on both increasing reversible electrochemical active sites/structural defects and expanding interlayer spacing, which endow the elaborate anode material with a high capacity of 456 mA h g−1 @ 0.1 A g−1, a desirable rate capability as high as 253.2 mA h g−1 @ 5 A g−1 for sodium-ion storage, and an excellent long-cycling performance with almost no decay after 5000 cycles @ 5 A g−1. Significantly, dynamic investigation indicates that the sodium-ion storage process and transfer kinetics are closely related to the covalently encapsulated sulfur chains. This work furnishes a new and systemic insight into the rational design of sodium-ion anode materials, in which the covalent sulfur chemistry may play a crucial role in reaching the supreme performance of sodium-ion batteries.


 Please wait while we load your content...
Please wait while we load your content...