The role of electrode wettability in electrochemical reduction of carbon dioxide†
Abstract
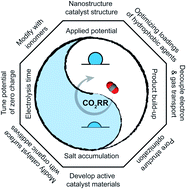
The electrochemical reduction of carbon dioxide (CO2RR) requires access to ample gaseous CO2 and liquid water to fuel reactions at high current densities for industrial-scale applications. Substantial improvement of the CO2RR rate has largely arisen from positioning the catalyst close to gas–liquid interfaces, such as in gas-diffusion electrodes. These requirements add complexity to an electrode design that no longer consists of only a catalyst but also a microporous and nanoporous network of gas–liquid–solid interfaces of the electrode. In this three-dimensional structure, electrode wettability plays a pivotal role in the CO2RR because the affinity of the electrode surface by water impacts the observed electrode reactivity, product selectivity, and long-term stability. All these performance metrics are critical in an industrial electrochemical process. This review provides an in-depth analysis of electrode wettability's role in achieving an efficient, selective, and stable CO2RR performance. We first discuss the underlying mechanisms of electrode wetting phenomena and the foreseen ideal wetting conditions for the CO2RR. Then we summarize recent advances in improving cathode performance by altering the wettability of the catalyst layer of gas-diffusion electrodes. We conclude the review by discussing the current challenges and opportunities to develop efficient and selective cathodes for CO2RR at industrially relevant rates. The insights generated from this review could also benefit the advancement of other critical electrochemical processes that involve multiple complex flows in porous electrodes, such as electrochemical reduction of carbon monoxide, oxygen, and nitrogen.
- This article is part of the themed collections: Journal of Materials Chemistry A Emerging Investigators and 2021 Journal of Materials Chemistry A most popular articles


 Please wait while we load your content...
Please wait while we load your content...