Resolving electron transfer kinetics in porous electrodes via diffusion-less cyclic voltammetry†
Abstract
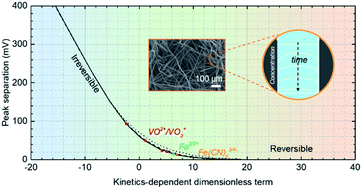
Porous electrodes are cornerstones in advanced electrochemical cells. They extend the electrode/electrolyte interface to three dimensions to boost reaction rates with high specific areas. The rate, however, depends on not only the specific area but also the electron transfer kinetics, the lack of characterization of which has hampered the design of better porous electrodes. The kinetics is often inferred from that on a planar electrode made of a similar material to avoid complications from mass transport in pores, despite likely discrepancies originating from structural and compositional differences due to the unique formation processes of the porous electrodes and their morphologies. Here we resolve electron transfer kinetics on commercial carbon felt, a type of indispensable yet poorly understood porous electrode for redox flow batteries, via a method of diffusion-less cyclic voltammetry. In this method, we place an active electrolyte inside the electrode and scan the voltammetry slowly, the results of which can be modelled accurately without invoking Fick's law and analyzed through a dimensionless term to derive the rate of electron transfer from the peak separation. Three RFB-relevant redox couples, Fe(CN)63/4−, Fe3/2+, and VO2+/VO2+, display rates in the range of 10−4 to 10−8 cm s−1, all much lower than those on glassy carbon. We confirm a correlation between the surface composition of the electrode and the reaction rate of VO2+/VO2+, corroborated by polarization curves attained in an all-vanadium RFB. The work offers a much-needed means towards the understanding of reaction mechanisms and better design of porous electrodes for and beyond RFBs.


 Please wait while we load your content...
Please wait while we load your content...