Synthesis, structure and diffusion pathways of fast lithium-ion conductors in the polymorphs α- and β-Li8SnP4†
Abstract
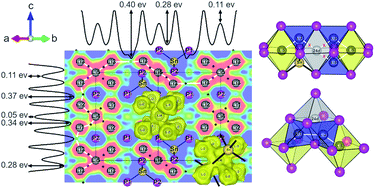
The increasing demand for a high-performance and low-cost battery technology promotes the search for Li+-conducting materials. Recently, phosphidotetrelates and -aluminates were introduced as an innovative class of phosphide-based Li+-conducting materials featuring ionic conductivities of up to 3 mS cm−1 at ambient temperature. In order to get a deeper understanding in structure–property relationship of lithium ion conductors closely related structures that differ in their ionic conductivity are of special interest. Here, we report on the two polymorphs α- and β-Li8SnP4, which show ionic conductivities of up to 0.7 mS cm−1 and low activation energies EA of about 28 kJ mol−1 (0.29 eV) at 298 K. The structures of the two phases are determined by single crystal X-ray and powder neutron diffraction experiments at different temperatures, and their significantly different ionic conductivities allow for a detailed insight into the structure–property relationship. The investigations are completed by 6Li, 31P and 119Sn solid state magic angle spinning NMR, temperature-dependent 7Li NMR experiments and electrochemical impedance spectroscopy. Negative nuclear density maps reconstructed from experimental structure factors were analyzed by the maximum entropy method (MEM) and the one-particle-potential (OPP) formalism. Distinct Li+ migration pathways including divergent activation barriers have been identified, which allow to interpret the different conductivities of the two modifications. The importance of partially occupied octahedral sites in the β-phase is ascertained to cause considerably lower energy barriers to adjacent tetrahedral voids, which promote the higher conductivity in comparison to the α-phase. The title compounds complete the series of three phosphidotetrelates (α-)Li8SiP4, α-Li8GeP4 and β-Li8GeP4, and allow a detailed investigation of the structure–property relationships for further tailoring of the material properties.


 Please wait while we load your content...
Please wait while we load your content...