The structural stability and defect-tolerance of ionic spinel semiconductors for high-efficiency solar cells†
Abstract
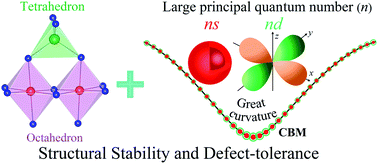
Incompatibility between defect-tolerance and structural stability is a severe issue hindering the wide application of high-efficiency solar cells. Usually, covalent/polar semiconductors with a prototype of Si/CdTe crystals exhibit great structural stability due to their compact tetrahedral structure, yet they present extremely poor defect-tolerance arising from the similar electronegativity of their component elements. In contrast, ionic semiconductors, such as perovskite series, always exhibit favorable electronic properties of intrinsic defects caused by the great disparity of electronegativity between anions and cations, but are structurally unstable because of the sparsely composed octahedral building blocks supported by large cations. Combining the stable framework of covalent semiconductors and benign defects of ionic compounds, we find that HgX2S4 (X = In, Sc and Y) spinel semiconductors possess both the merits. The tightly combined tetrahedral and octahedral building blocks ensure the structural stability and the band edge with an ionic characteristic, mainly dominated by Hg-6s/X-ns(d) for the conduction band minimum (CBM) and S-3p orbitals for the valence band maximum (VBM), make HgX2S4 defect-tolerant. The prominent downward bending of the CBM caused by spatially spreading Hg-6s/X-ns(d) orbitals with a large principal quantum number (n) not only induces a suitable optical band gap which is often too large in ionic compounds, but also promotes the formation and transport of n-type carriers. This study shows that Hg-based chalcogenide spinels are promising candidates for high-efficiency solar cells, and suggests that adopting cations with delocalized orbitals under the framework of spinel crystal is an alternative approach for synthesizing stable and defect-tolerant photovoltaic materials.


 Please wait while we load your content...
Please wait while we load your content...