Chemomechanical effect of reduced graphene oxide encapsulation on hydrogen storage performance of Pd nanoparticles†
Abstract
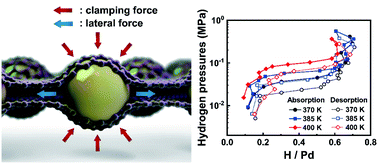
Primary chemomechanical impacts of confinement on hydrogen storage performance are studied using a nanolaminate structure where reduced graphene oxide (rGO) encapsulates palladium (Pd) nanoparticles. Three contributing factors are identified that can alter the reaction enthalpy: nanosizing, chemical interaction with the encapsulant, and mechanical stress induced strain from a combination of clamping force and lateral pulling force exerted on the Pd nanoparticles. The mechanical contributions are quantified by combining transmission electron microscopy, ab initio computation, and continuum elasticity theory, from which the encapsulation is found to exert an additional strain of 4.96% and 2.99% before and after hydrogen absorption, respectively, increasing the Pd and Pd hydride (PdHx) reaction enthalpy by 1.3–2.8 kJ (mol H2)−1. The effect of the chemical interaction with rGO also raises the reaction enthalpy by up to 1.6 kJ (mol H2)−1, while the nanosizing effect decreases the reaction enthalpy. The three contributing factors to the reaction enthalpy are found to be similar in magnitude, where the net effect is in agreement with the measured enthalpy increase of 3.7 kJ (mol H2)−1 from the bulk value. Hydrogen absorption kinetics and capacity also improved, which is attributed to facile nucleation of the hydrogen-rich phase enabled by the inhomogeneous strain distribution over the encapsulated PdHx nanoparticles. These results demonstrate that the chemomechanical effect can be controlled in the nanolaminate structure, providing an ideal template for tuning hydrogen storage performance.


 Please wait while we load your content...
Please wait while we load your content...