Facilitating electrocatalytic hydrogen evolution via multifunctional tungsten@tungsten disulfide core–shell nanospheres†
Abstract
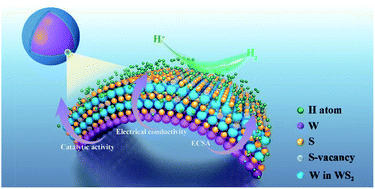
Transition metal dichalcogenides (TMDs) have long been thought to be alternatives for noble Pt/Ir metals as hydrogen evolution reaction electrocatalysts. However, sparse active sites and poor electrical property, two of the most formidable challenges for their electrocatalytic performance, can hardly be improved just by existing methods. Herein, tungsten (W)@tungsten disulfide (WS2) core–shell nanospheres (CSNSs) were synthesized via one-step laser-assisted strategy. Through introducing multifunctional CSNSs, metallic WS2 hierarchically curves on the W core with a curvature as high as 10−2 nm−1 make the interlayer spacing of WS2 broaden significantly and then expose more catalytic sites. Moreover, the electrical conductivity of the electrocatalyst rises 4.5 times via a highly electroconductive W core. The electrochemical surface area of WS2 is also greatly enlarged by forming a 3D nanospheres-aggregated nanoporous van der Waals heterostructure. Calculations indicate that CSNSs contribute to optimizing the hydrogen adsorption Gibbs free energy, and modulating the electronic occupation around the Fermi level. Among these, W sites on the curved shell emerge as the most active catalytic centers. Further experiments demonstrate that W@WS2 CSNSs can therefore exhibit superior catalytic activities featuring a small overpotential of 161 mV at 10 mA cm−2, an ultralong durability in acids (∼100 h) and most importantly, the smallest Tafel slope (34.5 mV dec−1) and the biggest electrical double-layer capacitance (62.2 mF cm−2) among WS2-based electrocatalysts. This study opens a pathway to develop site-rich and highly electroconductive TMDs, which not only act as high performance water electrolysis catalysts on a large scale, but can also be used for batteries, supercapacitors and electrochemical actuators.


 Please wait while we load your content...
Please wait while we load your content...