A non-flammable electrolyte for long-life lithium ion batteries operating over a wide-temperature range†
Abstract
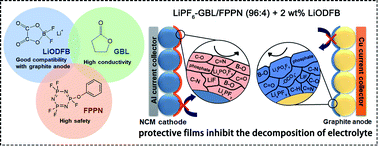
(Phenoxy)pentafluorocyclotriphosphazene (FPPN), a kind of flame retardant, is initially mixed with gamma-butyrolactone (GBL) to formulate a new safe electrolyte, in which lithium difluoro(oxalato)borate (LiODFB) is used as the additive to improve the electrode interfacial properties. It is found that the addition of FPPN ensures that this electrolyte is totally non-flammable, and the conductivity of this electrolyte is about 10 mS cm−1, which is equivalent to that of most commercial electrolytes. More significant, this high safety electrolyte helps graphite/LiNi0.5Co0.2Mn0.3O2 full cells to obtain preeminent electrochemical performance. The capacity retention of the cells reaches 85.4% after 500 cycles at 1C and room temperature, which is apparently better than that of a commercial electrolyte. Its cycle performance at elevated temperature (60 °C) exceeds that of a commercial electrolyte. Furthermore, the cell with this electrolyte can deliver a discharge capacity of about 90 mA h g−1 at low temperature (−40 °C). To sum up, such an electrolyte can ensure safety and electrochemical performance in a wide-temperature range of operating conditions. The interfacial properties of electrodes are also tested by XPS and SEM measurements, and this is the first time to analyze the influence of fluorinated cyclophosphazenes on the graphite anode.


 Please wait while we load your content...
Please wait while we load your content...