Self-supported ultra-active NiO-based electrocatalysts for the oxygen evolution reaction by solution combustion†
Abstract
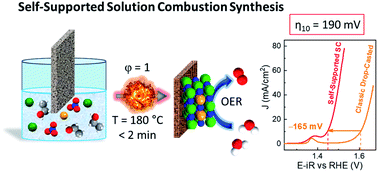
The oxygen evolution reaction (OER) is a fundamental process to develop a technology that can drive energy transition towards renewable and sustainable fuels. Nevertheless, efficient and straightforward methodologies to obtain superior and stable electrodes need to be implemented to approach this technology to real applications. Recently, self-supported catalysis emerged as a promising solution. However, catalyst design is still limited by the low chemical tunability and elevated preparation times and costs. Herein, a solution combustion (SC) methodology is described to produce designed self-supported electrocatalysts that excel in the OER and mitigate previous limitations. M-doped NiO-based electrocatalysts (with M = Fe, Co, Mn, and Zn) were self-supported by the SC method on nickel foam, and overperformed analogous benchmarked catalysts prepared by other methods. Notably, in Fe-doped NiO, the overpotential required to drive the OER at 10 mA cm−2 was found to be 190 mV, the lowest reported so far for metal oxide electrocatalysts at pH 13. By the combination of spectroelectrochemical (SEC) and electrochemical impedance spectroscopy (EIS), we studied the role of the metal dopant cation, showing that dopant metals assist the formation of the active species responsible for the high (electro)catalytic activity. We envision that the presented simple, cost-time efficient methodology would stimulate the preparation and study of effective self-supported metal-oxide catalysts for a broad range of applications.


 Please wait while we load your content...
Please wait while we load your content...