Highly efficient atomically dispersed Co–N active sites in porous carbon for high-performance capacitive desalination of brackish water†
Abstract
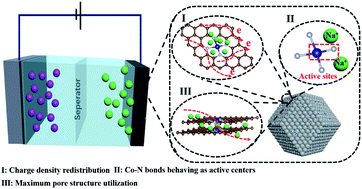
Carbon materials with external defects (heteroatoms) have been widely studied to promote their performance in capacitive deionization (CDI), but the salt adsorption capacity and rate capability are far from meeting the rapid demand for clean water. Further, the origin of capacitive activity from the external defects, especially the nitrogen-coordinated transition metal sites in carbons, cannot be clearly clarified. Herein, porous carbons with highly efficient atomically dispersed Co–N active sites are designed and prepared by a one-step scalable pyrolysis process from a binary metal–organic framework. The optimized carbon material exhibits an excellent specific capacitance of 229 F g−1 at 2 mV s−1 in a 1.0 M NaCl solution. Further, the salt adsorption capacity is as high as 55.6 mg g−1 in 1000 mg L−1 NaCl electrolyte at 1.2 V. The experimental analysis reveals that the presence of atomically dispersed Co–N bonds as effective active sites is confirmed to play a crucial role in the CDI performance, as they are beneficial for the ion adsorption behavior and maximize the utilization of the porous structure. Furthermore, theoretical simulations demonstrate that the highly efficient Co–N moieties as the active centers can remarkably facilitate charge density redistribution, thereby improving the intrinsic affinity of ions and thus fundamentally enhancing the CDI performance. This work affords a deep insight for further understanding the critical role of Co–N sites in carbon-based CDI performance.


 Please wait while we load your content...
Please wait while we load your content...