Enhanced N2 affinity of 1T-MoS2 with a unique pseudo-six-membered ring consisting of N–Li–S–Mo–S–Mo for high ambient ammonia electrosynthesis performance†
Abstract
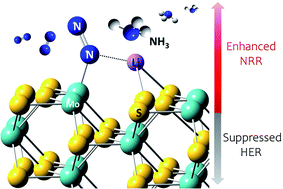
The Haber–Bosch process is widely used to convert atmospheric nitrogen (N2) into ammonia (NH3). However, the extreme reaction conditions and abundant carbon released by this process make it important to develop a greener NH3 production method. The electrochemical nitrogen reduction reaction (NRR) is an attractive alternative to the Haber–Bosch process. Herein, we demonstrated that molybdenum sulfide on nickel foil (1T-MoS2–Ni) with low crystallinity was an active NRR electrocatalyst. 1T-MoS2–Ni achieved a high faradaic efficiency of 27.66% for the NRR at −0.3 V (vs. RHE) in a LiClO4 electrolyte. In situ X-ray diffraction and ex situ X-ray photoemission analyses showed that lithium ions were intercalated into the 1T-MoS2 layers during the NRR. Moreover, theoretical calculations revealed the differences between six membered rings formed in the 1T-MoS2 and 2H-MoS2 systems with Li intercalation. The bond distances of d(Mo–N) and d(N–Li) of in Li–1T-MoS2 were found to be shorter than those in Li–2H-MoS2, resulting in a lower energy barrier of N2 fixation and higher NRR activity. Therefore, 1T-MoS2–Ni is promising as a scalable and low-cost NRR electrocatalyst with lower power consumption and carbon emission than the Haber–Bosch process.


 Please wait while we load your content...
Please wait while we load your content...