Polymer-inspired covalent triazine frameworks from the carbonaceous side – influence of unexpected surface functionalisation on liquid-phase adsorption processes†
Abstract
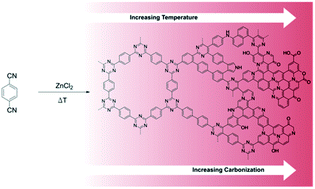
The selective liquid-phase adsorption represents a highly efficient and sustainable alternative to common separation techniques in biorefinery processes. A variety of covalent triazine frameworks (CTFs) have been scrutinized as adsorbents for the selective separation of biorefinery products such as itaconic acid and lysine from glucose-based mixtures. Owing to the tunability of the framework's properties by means of a judicious choice of monomers and synthesis route, structure–adsorption-relationships for the compounds of interest could be derived. Detailed characterisation based on N2-physisorption, H2O-vapor sorption, elemental analysis, XPS, (CO2)-TPD-MS, FT-IR, NMR and ICP-OES have served to shed light on the composition of this established class of materials while emphasizing their carbonaceous nature. For itaconic acid–glucose mixtures more hydrophobic materials, based on 4,4′-dicyanobiphenyl showed the best separation factors while for lysine–glucose mixtures high hydrophilicity was beneficial for lysine adsorption. The use of phosphorous pentoxide instead of the traditional ZnCl2 for the synthesis of CTFs gave rise to an unexpected functionalisation with phosphorous and oxygen-containing groups whose presence was found to markedly improve the CTF's separation performance for lysine from glucose mixtures. The distribution of phosphorous linkages present at the surface varied significantly with synthesis temperature, which was elucidated by XPS, 31P MAS NMR and TPD-MS experiments.


 Please wait while we load your content...
Please wait while we load your content...