Unprecedented CO2 adsorption behaviour by 5A-type zeolite discovered in lower pressure region and at 300 K†
Abstract
Mitigation of the amounts of CO2 in the environment is one of the most urgent problems requiring a solution. To fulfil this demand, efficient adsorbents for CO2 are required that work at room temperature (RT) and in a lower pressure region of not more than 5000 ppm under ambient conditions. In the present work, specific and selective adsorption of CO2 onto NaCaA-85 (A-type zeolite with an ion-exchange capacity of 85%) was observed under the required conditions; the amounts of adsorbed CO2 on the NaCaA-85 sample were far larger than amounts reported for other materials in the pressure range from 400 to 5000 ppm. The characteristic adsorption mechanism induced by this material was verified directly through methods combining synchrotron-based far-infrared (far-IR) measurements with a computational technique. The resultant Ca2+-framework vibration modes for the NaCaA-85 sample were observed at 266 and 246 cm−1, which shifted toward the lower wave-numbers, i.e., 225 and 203 cm−1, after CO2 adsorption at RT, respectively. The observed characteristic property was explained by a model consisting of a CO2 molecule simultaneously pinned by two Ca2+ ions positioned on two types of exchangeable sites composed of 8- and 6-membered rings, which was well supported by the density functional theory calculation method. This characteristically adsorbed CO2 species was completely desorbed, and the original state easily recovered through evacuation around 400 K. In addition, the selective adsorption behaviour of CO2 from other gases, such as H2, CH4, O2 and N2, was found at RT. On the basis of these data, the separation properties of CO2 were examined by measuring the breakthrough curve using a model gas composed of 0.04% CO2, 20% O2 and 79.96% N2, which mimicked ambient air, indicating the superior separation feature. These findings may pave a new way for the use of the NaCaA-85 material as an efficient adsorbent for selective CO2 adsorption functioning at RT and in the lower pressure region of up to 5000 ppm.
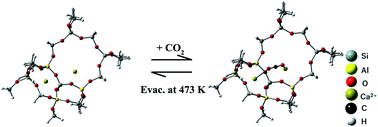


 Please wait while we load your content...
Please wait while we load your content...