Self-assembled luminescent cholate gels induced by a europium ion in deep eutectic solvents†
Abstract
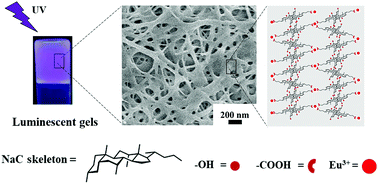
Deep eutectic solvents (DESs) with excellent physicochemical properties similar to ionic liquids and biocompatibility are potential solvent candidates for designing novel lanthanide luminescent soft materials. In this paper, the fabrication and characterization of such luminescent gels in three choline chloride (ChCl)-based DESs through self-assembly of the sodium cholate and europium nitrate are presented. The microstructure and gel-like nature of the obtained eutectogels were explored and confirmed by scanning electron microscopy and rheology measurements. While Fourier transform infrared spectroscopy and small-angle X-ray scattering were used to analyze the gel formation mechanism, which was considered to be synergistically driven by metal coordination, hydrogen bonding and solvophobic interactions. All three eutectogels exhibited good photophysical properties. Among these, the one formed in ChCl/urea DES was found to possess the strongest mechanical strength. While the one formed in ChCl/glycerol DES exhibited the longest luminescence lifetime and quantum efficiency. The obtained results demonstrate the possibility of using DESs to construct lanthanide luminescent soft materials or control their properties through the choice of hydrogen-bond donor molecules.


 Please wait while we load your content...
Please wait while we load your content...