Sm0.5Sr0.5Fe0.8M0.2O3−δ (M = Co, Cu) perovskite oxides for efficient oxygen evolution reaction in alkaline electrolyte†
Abstract
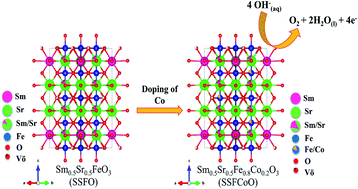
Development of an efficient, earth-abundant, inexpensive, and stable perovskite electrocatalyst to replace RuO2 catalysts for the oxygen evolution reaction (OER) is much needed for the practical application of electrochemical energy conversion devices. Herein, we report Sm0.5Sr0.5Fe0.8M0.2O3−δ (M = Co, Cu) perovskite doped with different transition metals at the B-site as a cheaper OER electrocatalyst in 1 M KOH electrolyte. Among the prepared catalysts, Sm0.5Sr0.5Fe0.8Co0.2O3−δ (SSFCoO) catalysts show improved OER activity with an on-set potential of 1.48 V vs. RHE with a lower overpotential of 316 mV at 10 mA cm−2. Furthermore, the OER performance correlated with the electronic structure of the electrocatalyst was analyzed through X-ray photoelectron spectra which revealed that the improved OER activity of the SSFCoO catalyst is due to an increased Fe4+ oxidization state. Strong Fe 3d–O 2p hybridization causes up-shifting of the O 2p valence band close to the Fermi level from 1.0 eV to 0.33 eV, significantly reducing the charge transfer energy and causing faster OH− adsorption and desorption for efficient OER activity.


 Please wait while we load your content...
Please wait while we load your content...