A Paal–Knorr agent for chemoproteomic profiling of targets of isoketals in cells†
Abstract
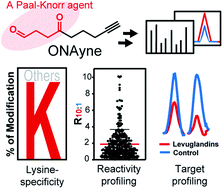
Natural systems produce various γ-dicarbonyl-bearing compounds that can covalently modify lysine in protein targets via the classic Paal–Knorr reaction. Among them is a unique class of lipid-derived electrophiles – isoketals that exhibit high chemical reactivity and critical biological functions. However, their target selectivity and profiles in complex proteomes remain unknown. Here we report a Paal–Knorr agent, 4-oxonon-8-ynal (herein termed ONAyne), for surveying the reactivity and selectivity of the γ-dicarbonyl warhead in biological systems. Using an unbiased open-search strategy, we demonstrated the lysine specificity of ONAyne on a proteome-wide scale and characterized six probe-derived modifications, including the initial pyrrole adduct and its oxidative products (i.e., lactam and hydroxylactam adducts), an enlactam adduct from dehydration of hydroxylactam, and two chemotypes formed in the presence of endogenous formaldehyde (i.e., fulvene and aldehyde adducts). Furthermore, combined with quantitative chemoproteomics in a competitive format, ONAyne permitted global, in situ, and site-specific profiling of targeted lysine residues of two specific isomers of isoketals, levuglandin (LG) D2 and E2. The functional analyses reveal that LG-derived adduction drives inhibition of malate dehydrogenase MDH2 and exhibits a crosstalk with two epigenetic marks on histone H2B in macrophages. Our approach should be broadly useful for target profiling of bioactive γ-dicarbonyls in diverse biological contexts.


 Please wait while we load your content...
Please wait while we load your content...