Using fluorene to lock electronically active moieties in thermally activated delayed fluorescence emitters for high-performance non-doped organic light-emitting diodes with suppressed roll-off†
Abstract
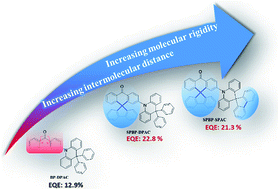
Thermally activated delayed fluorescence (TADF) emitters with aggregation-induced emission (AIE) features are hot candidates for non-doped organic light-emitting diodes (OLEDs), as they are highly emissive in solid states upon photoexcitation. Nevertheless, not every AIE-TADF emitter in the past had guaranteed decent efficiencies in non-doped devices, indicating that the AIE character alone does not necessarily afford ideal non-doped TADF emitters. As intermolecular electron-exchange interaction that involves long-lived triplet excitons plays a dominant role in the whole quenching process of TADF, we anticipate that it is the main reason for the different electroluminescence performances of AIE-TADF emitters. Therefore, in this work, we designed two TADF emitters SPBP-DPAC and SPBP-SPAC by modifying a reported less successful emitter BP-DPAC with extra fluorenes to increase intermolecular distances and attenuate this electron-exchange interaction. With the fluorene lock as steric hindrance, SPBP-DPAC and SPBP-SPAC exhibit significantly higher exciton utilization in non-doped films due to the suppressed concentration quenching. The non-doped OLEDs based on SPBP-DPAC and SPBP-SPAC show an excellent maximum external quantum efficiency (EQE) of 22.8% and 21.3% respectively, and what's even more promising is that ignorable roll-offs at practical brightness (e.g., 1000 and 5000 cd m−2) were realized. These results reveal that locking the phenyl rings as steric hindrance can not only enhance the molecular rigidity, but also cause immediate relief of concentration quenching, and result in significant performance improvement under non-doped conditions. Our approach proposes a feasible molecular modification strategy for AIE-TADF emitters, potentially increasing their applicability in OLEDs.


 Please wait while we load your content...
Please wait while we load your content...