Reversible carbon–boron bond formation at platinum centers through σ-BH complexes†
Abstract
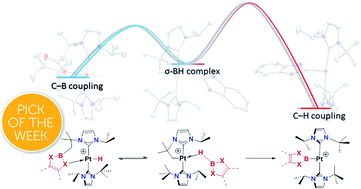
A reversible carbon–boron bond formation has been observed in the reaction of the coordinatively unsaturated, cyclometalated, Pt(II) complex [Pt(ItBuiPr′)(ItBuiPr)][BArF], 1, with tricoordinated boranes HBR2. X-ray diffraction studies provided structural snapshots of the sequence of reactions involved in the process. At low temperature, we observed the initial formation of the unprecedented σ-BH complexes [Pt(HBR2)(ItBuiPr′)(ItBuiPr)][BArF], one of which has been isolated. From −15 to +10 °C, the σ-BH species undergo a carbon–boron coupling process leading to the platinum hydride derivative [Pt(H)(ItBuiPr–BR2)(ItBuiPr)][BArF], 4. Surprisingly, these compounds are thermally unstable undergoing carbon–boron bond cleavage at room temperature that results in the 14-electron Pt(II) boryl species [Pt(BR2)(ItBuiPr)2][BArF], 2. This unusual reaction process has been corroborated by computational methods, which indicate that the carbon–boron coupling products 4 are formed under kinetic control whereas the platinum boryl species 2, arising from competitive C–H bond coupling, are thermodynamically more stable. These findings provide valuable information about the factors governing productive carbon–boron coupling reactions at transition metal centers.
- This article is part of the themed collections: Spotlighting main group elements in polynuclear complexes, 2020 ChemSci Pick of the Week Collection and 2020 Chemical Science HOT Article Collection


 Please wait while we load your content...
Please wait while we load your content...