Highly enhanced electrical properties of lanthanum-silicate-oxide-based SOFC electrolytes with co-doped tin and bismuth in La9.33−xBixSi6−ySnyO26
Abstract
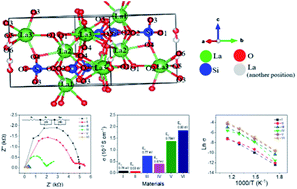
Solid oxide fuel cells (SOFCs) are one of the most promising clean energy sources to be developed. However, the operating temperature of SOFCs is currently still very high, ranging between 1073 and 1273 K. Reducing the operating temperature of SOFCs to intermediate temperatures in between 773 and 1073 K without decreasing the conductivity value is a challenging research topic and has received much attention from researchers. The electrolyte is one of the components in SOFCs which has an important role in reducing the operating temperature of the SOFC compared to the other two fuel cell components, namely the anode and cathode. Therefore, an electrolyte that has high conductivity at moderate operating temperature is needed to obtain SOFC with medium operating temperature as well. La9.33Si6O26 (LSO) is a potential electrolyte that has high conductivity at moderate operating temperatures when this material is modified by doping with metal ions. Here, we report a modification of the structure of the LSO by partial substitution of La with Bi3+ ions and Si with Sn4+, which forms La9.33−xBixSi6−ySnyO26 with x = 0.5, 1.0, 1.5, and y = 0.1, 0.3, 0.5, in order to obtain an electrolyte of LSO with high conductivity at moderate operating temperatures. The addition of Bi and Sn as dopants has increased the conductivity of the LSO. Our work indicated highly enhanced electrical properties of La7.83Bi1.5Si5.7Sn0.3O26 at 873 K (1.84 × 10−2 S cm−1) with considerably low activation energy (Ea) of 0.80 eV comparing to pristine La9.33Si6O26 (0.08 × 10−2 S cm−1).


 Please wait while we load your content...
Please wait while we load your content...