Hierarchical NaFePO4 nanostructures in combination with an optimized carbon-based electrode to achieve advanced aqueous Na-ion supercapacitors†
Abstract
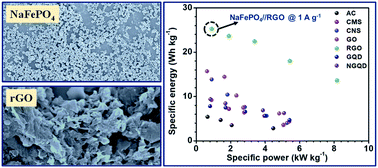
Recent trends in sodium-ion-based energy storage devices have shown the potential use of hollow structures as an electrode material to improve the performance of these storage systems. It is shown that, in addition to the use of hierarchical structures, the choice of the complementary carbon electrode determines the final performance of Na-ion-based devices. Here, we present simple synthesis strategies to prepare different structured carbonaceous materials that can be upscaled to an industrial level. Individual carbon materials deliver specific capacitance ranges from 120 to 220 F g−1 at a current density of 1 A g−1 (with excellent capacity retention). These structures, when combined with hollow NaFePO4 microspheres to fabricate an aqueous supercapacitor, show as high as a 1.7 V working potential window and can deliver a maximum energy density of 25.29 W h kg−1 capacity retention. These values are much higher than those reported by NaFePO4 solid particles and randomly chosen carbon structure-based supercapacitors.


 Please wait while we load your content...
Please wait while we load your content...