Molecular mechanism of direct electron transfer in the robust cytochrome-functionalised graphene nanosystem†
Abstract
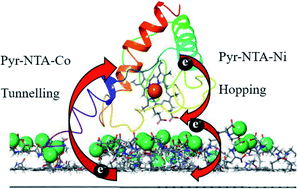
Construction of green nanodevices characterised by excellent long-term performance remains high priority in biotechnology and medicine. Tight electronic coupling of proteins to electrodes is essential for efficient direct electron transfer (DET) across the bio-organic interface. Rational modulation of this coupling depends on in-depth understanding of the intricate properties of interfacial DET. Here, we dissect the molecular mechanism of DET in a hybrid nanodevice in which a model electroactive protein, cytochrome c553 (cyt c553), naturally interacting with photosystem I, was interfaced with single layer graphene (SLG) via the conductive self-assembled monolayer (SAM) formed by pyrene–nitrilotriacetic acid (pyr–NTA) molecules chelated to transition metal redox centers. We demonstrate that efficient DET occurs between graphene and cyt c553 whose kinetics and directionality depends on the metal incorporated into the bio-organic interface: Co enhances the cathodic current from SLG to haem, whereas Ni exerts the opposite effect. QM/MM simulations yield the mechanistic model of interfacial DET based on either tunnelling or hopping of electrons between graphene, pyr–NTA–M2+ SAM and cyt c553 depending on the metal in SAM. Considerably different electronic configurations were identified for the interfacial metal redox centers: a closed-shell system for Ni and a radical system for the Co with altered occupancy of HOMO/LUMO levels. The feasibility of fine-tuning the electronic properties of the bio-molecular SAM upon incorporation of various metal centers paves the way for the rational design of the optimal molecular interface between abiotic and biotic components of the viable green hybrid devices, e.g. solar cells, optoelectronic nanosystems and solar-to-fuel assemblies.


 Please wait while we load your content...
Please wait while we load your content...