Identification of (2R,3R)-2-(3,4-dihydroxyphenyl)chroman-3-yl-3,4,5-trihydroxy benzoate as multiple inhibitors of SARS-CoV-2 targets; a systematic molecular modelling approach†
Abstract
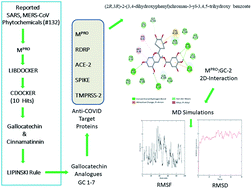
Coronavirus disease of 2019 (COVID-19) is a zoonotic disease caused by a new severe acute respiratory syndrome (SARS-CoV-2) which has quickly resulted in a pandemic. Recent anti-COVID-19 drug discoveries are leaning towards repurposing phytochemicals which have been previously reported for SARS and MERS-CoV outbreaks. However, they have been either virtually screened or tested so far against mono targets and the potent derivatives of virtually sorted lead molecules remain elusive. We aimed to identify the phytochemicals having potentials to inhibit SARS CoV-2 infection via multiple targets. The selected 132 phytochemicals were virtually screened using a structure based in silico technique against main protease (Mpro) which is a potential target of SARS CoV-2. Six compounds were selected based on the LibDock scores and further subjected to induced fit docking using the CDOCKER module of DS. Two compounds namely cinnamtannin-B and gallocatechin gallate were identified as top HITS against main protease (Mpro). Based on the Lipinski rule of five (L-ROF) and synthetic feasibility, gallocatechin gallate was taken for our further studies. Six analogues of gallocatechin gallate were screened against the next important targets such as RNA-dependent RNA polymerase (RdRp), angiotensin converting enzyme-2 (ACE2), transmembrane protease serine -2 (TMPRSS2) and interleukin-6 (IL-6) along with main protease (Mpro). Our molecular docking results reveal that a gallocatechin analogue (GC-2) namely (2R,3R)-2-(3,4-dihydroxyphenyl)chroman-3-yl-3,4,5-trihydroxy benzoate has shown potential to inhibit multiple targets of SARS CoV-2. Further, the molecular dynamics study was carried out to ascertain the stability of the GC-2 and RdRp complex.
- This article is part of the themed collections: Coronavirus articles - free to access collection and 2021 RSC Advances HOT Article Collection


 Please wait while we load your content...
Please wait while we load your content...