Facile mass preparation and characterization of Al/copper ferrites metastable intermolecular energetic nanocomposites†
Abstract
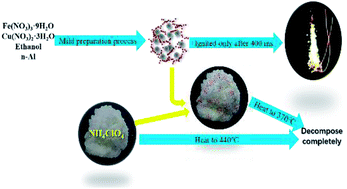
In the present work, a novel Al/copper ferrites metastable intermolecular energetic nanocomposite was prepared by a simple and mild sol–gel method followed by low temperature calcination, and characterized by various analytical techniques. The X-ray diffraction (XRD) analysis suggests that the products contain crystal forms of aluminum and spinel-type ferrite crystal forms which are CuFe2O4 with many crystal defects. The scanning electron microscopy (SEM) and nitrogen adsorption–desorption analyses reveal that the prepared Al/copper ferrites are mesoporous structures with large specific surface areas of up to 184.47 m2 g−1 and further reveal the pore construction of this material. Its crystal defects and large specific surface area provide the possibility for its excellent catalytic performance. Al/copper ferrites have 45% better exothermic properties with higher energy output efficiency, faster burning rate, and higher reactivity than traditional Al/Fe2O3 prepared by the same method. Due to the synergistic catalytic effect of Cu–Fe oxides, Al/copper ferrites have a better catalytic effect on AP thermal decomposition and can reduce the HTD peak temperature of AP 33% more than Al/Fe2O3. The catalytic mechanism of Al/copper ferrites for the thermal decomposition of AP is obtained based on the electron transfer theories, synergistic catalytic mechanism, and the porous structure of Al/copper ferrites. Due to the mild reaction conditions and low calcination temperature, dozens of grams of product can be safely obtained at one time with low cost and easily available raw materials to meet the requirements of propellant up to several kilograms or other industrial applications.


 Please wait while we load your content...
Please wait while we load your content...